- Blog
- Gin rummy 500 rules
- Idle oil tycoon efficiency manager
- Keywe xbox
- Bright memory infinite xbox game pass
- The missing link hip and joint reviews
- Alan wake 2 tunnels in coal mine
- Computer work timer
- Tits wiki offline
- Flight simulator x landings
- Periodic table f
- Public radio international jingle
- Ancestry world explorer plus coupon code
- Displaylink usb graphics adapters and docs driver
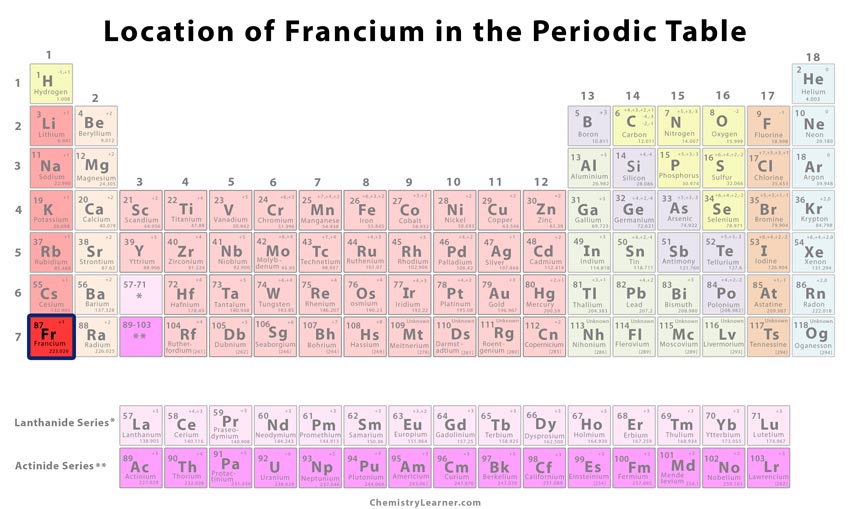
The gases of zero group (Noble gases) are unreactive because they have a stable electronic configuration. They consists of two Horizontal rows of metals at the bottom of the table. Inner transition elements: Inner transition elements are the elements belonging to Group 3 in 6th and 7th period. Transition elements consists of metals lying between strongly elctropositive metals on a left and least electropositive elements on the right. Transition elements – Elements belonging to Group 3 to 12 are called transition elements. State why noble gases are considered unreactive elements. State the position of the inner transition elements. What are transition elements and inner transition elements. (d) Group 13-16 – Post Transition elements. (c) Group-3 to 12 IB to VII B to VIII – Transition elements- metals. These vertical columns are called Groups.It signifies the same electronic configuration and similar properties. There are 18 vertical columns in the Modem periodic table. What are ‘groups’ of the Modern Periodic Table ? What does the ‘group number’ signify.
the bonding and character of oxides of period-3 – group 1, 13 and 16. the bonding and state of chlorides of period-3 – group 1, 15, 16 and Electronegativity – increases (with the exception of Noble gases).Metallic character decreases while non-metallic character increases (with the exception of Noble gases).Number of valence electrons increases by one.The property trends in general on moving from left to right in a period of the periodic table are:

State the property trends in general on moving from left to right in a period of the periodic table. Li, Be, B, C are called Bridge elements because they show similarties in properties diagonally with the period of the next group. (b) period-2 elements are ‘called bridge elements’.Ĭompletion of each period is logical since each period begins with Group 1 element having one electron in outermost shell and ends with Group 18 element having filled outermost shell with 8 electrons.
- Blog
- Gin rummy 500 rules
- Idle oil tycoon efficiency manager
- Keywe xbox
- Bright memory infinite xbox game pass
- The missing link hip and joint reviews
- Alan wake 2 tunnels in coal mine
- Computer work timer
- Tits wiki offline
- Flight simulator x landings
- Periodic table f
- Public radio international jingle
- Ancestry world explorer plus coupon code
- Displaylink usb graphics adapters and docs driver
